|
1/8/2024 0 Comments Anode cathode reaction writing 
In between these electrodes is the electrolyte, which contains ions that can freely move. Electrodes can be made from any sufficiently conductive materials, such as metals, semiconductors, graphite, and even conductive polymers. The cathode is the electrode where reduction takes place. The anode is defined as the electrode where oxidation occurs. These scientists conducted several experiments on chemical reactions and electric current during the late 18th century.Įlectrochemical cells have two conductive electrodes, called the anode and the cathode. This kind of cell includes the galvanic, or voltaic, cell, named after Luigi Galvani and Alessandro Volta. voltaic cell: A cell, such as in a battery, in which an irreversible chemical reaction generates electricity a cell that cannot be recharged.Īn electrochemical cell is a device that produces an electric current from energy released by a spontaneous redox reaction.half-cell: Either of the two parts of an electrochemical cell containing an electrode and an electrolyte.redox: A reversible chemical reaction in which one reaction is an oxidation and the reverse is a reduction.The half-cells are connected by a salt bridge that allows the ions in the solution to move from one half-cell to the other, so that the reaction can continue. 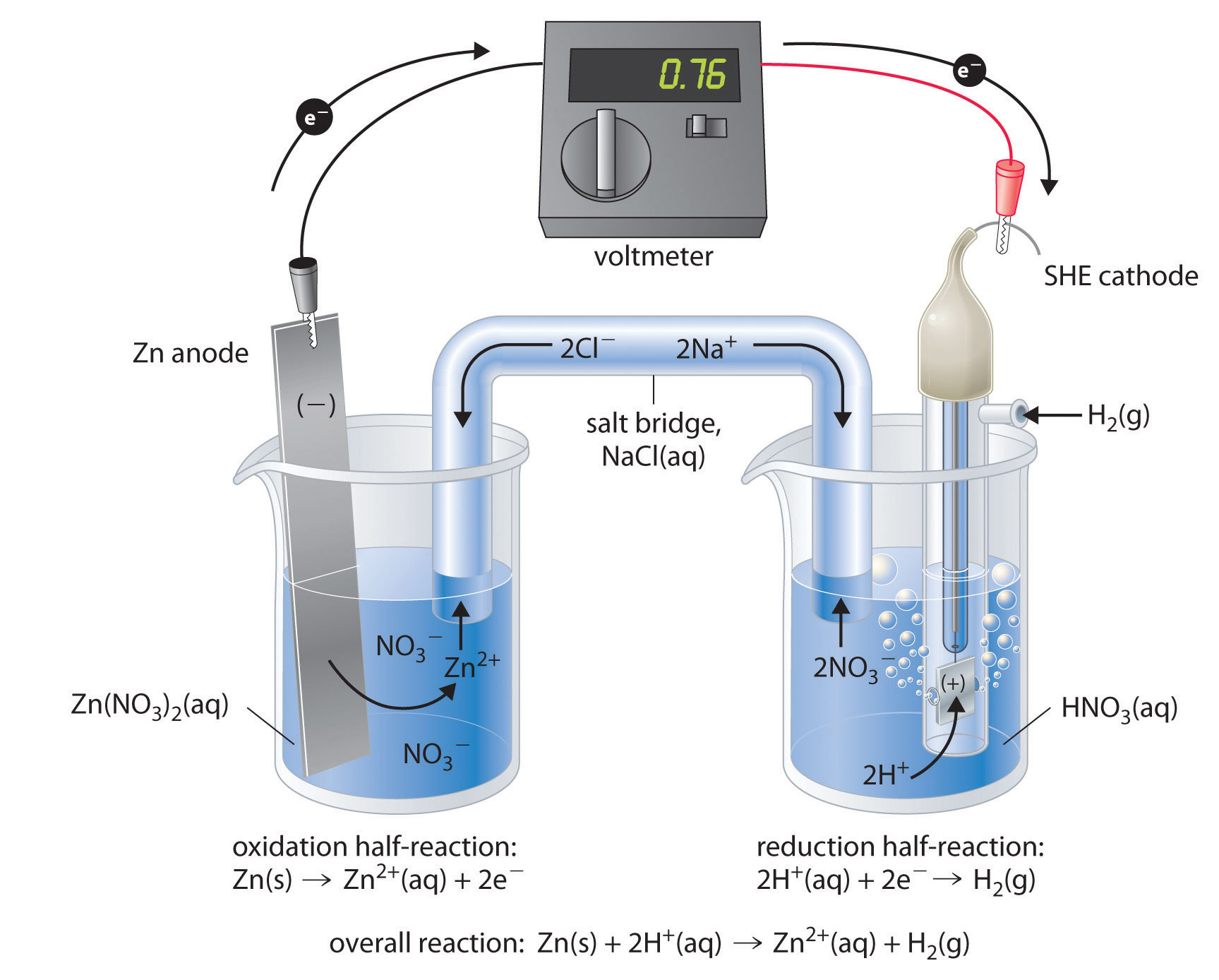
The electrons always flow from the anode to the cathode.Reduction describes the gain of electrons by a molecule, atom, or ion.Oxidation describes the loss of electrons by a molecule, atom, or ion. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
